COVID-19 Vaccine Will Take At Least 1 Year: EU Medicines Agency
especiales
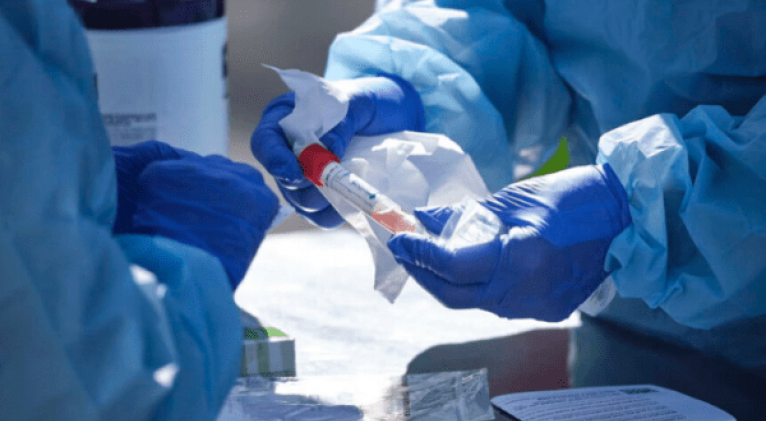
Human trials will begin imminently, but even if they go well and a cure is found, it will take at least a year for the vaccine to be effective worldwide.
The development of a vaccine against the new coronavirus will take at least a year, the European Medicines Agency (EMA) said Tuesday.
According to the EMA, it could be "at least a year before a COVID-19 vaccine is ready for approval and there are sufficient quantities to allow widespread use" worldwide.
The Amsterdam-based agency explained that it was based on current data and the time it took to develop other vaccines.
In addition, it added that two vaccines have already entered the first phase of clinical trials, which were being carried out with healthy volunteers.
But, in general, "the terms for the development of medicinal products are difficult to foresee," the agency added.
In fact, so far, no drug has been profiled as a fully effective treatment against the new coronavirus, which has so far claimed at least 40,000 lives.
According to data from the AFP news agency, the number of declared cases of COVID-19 worldwide exceeded 800,000 on Tuesday, so scientists are in a race against the clock to find a vaccine against the new virus that appeared in China at the end of last year.













Add new comment