CECMED authorizes to use phase-II trial with CIGB2020 candidate
especiales
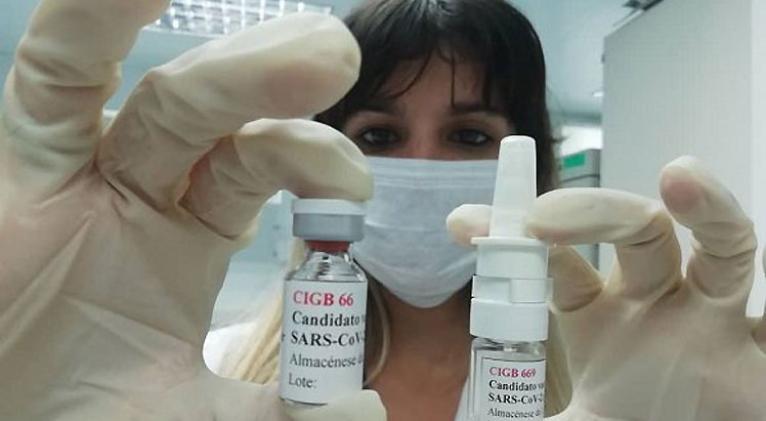
Cuba´s Center for State Control of medicines, Equipment and Medical Devices (CECMED) on Wednesday authorized the start of a phase-II clinical trial with the vaccine candidate CIGB2020, seeking to stimulate the immune response against acute respiratory infections.
The news was announced on Twitter by Dr. Gerardo Guillén, director of biomedical research at the Center for Genetic Engineering and Biotechnology (CIGB).
According to the source, ¨the study seeks to evaluate the effect and safety of CIGB2020 in subjects aged 60 years of age or older, close contacts of patients diagnosed with acute respiratory infections, compared with a control group of the same population.¨
CIGB2020 is a product that is applied nasal and sublingual to stimulate immunity at the local level, which is the entry of the virus into the body. The product has been effective in its application to individuals confirmed with Covid-19, limiting the progress of the condition to stages of greater complexity and severity.
The new product prevents the virus from overriding the body's immune system and, at the same time, attains a balance in natural or innate immunity, which contributes to saving lives and other remarkable benefits in risk groups.
It is an open and controlled study, and that a phase-I clinical trial had previously been carried out at the Naval Hospital of Havana, in people with respiratory symptoms, suspected of suffering from an acute infection, in particular with SARS-CoV-2.













Add new comment